Fine Chemicals & Intermediates
DAIKIN Industries produces and offers wide ranged intermediates through an integrated production system that starts with hydrofluoric acid.
Applications of fluorinated compounds, especially fluorinated intermediates are rapidly expanding and used for final products in many Life Science and Industrial applications.
Beyond that, Daikin Industries provides a series of fluorinating agents called „MEC Reagents“ as well as custom synthesis of various fluorochemicals using F2, IF5/Et3N-3HF, and SF4 that can efficiently fluorinate hydroxyl-, carbonyl- and carboxyl- groups, and is particularly suited to selective Difluorination (C=O into CF2).
MEC reagents are safe fluorinating agents for synthesis of new fluoro-compounds and are particularly useful for laboratory and bulk synthesis.
Intermediates
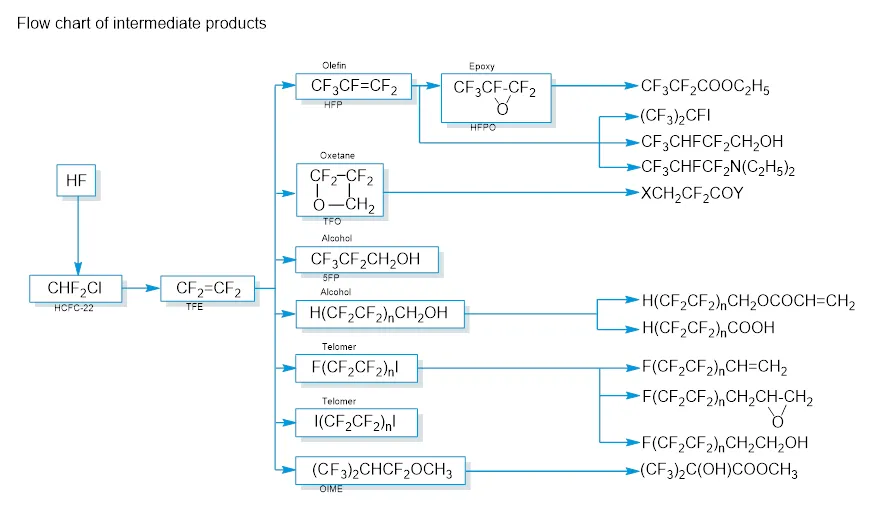
Daikin produces and offers a wide range of intermediates. The selection below provides a first impression of our product range and capabilities.
By combining various organic synthesis technologies, Daikin can propose an extensive selection of intermediates such as alcohols, acrylates, carboxylic acids, esters, epoxides, ethers, olefins, amines, iodides, methacrylates and others.
Fluorination Technology
Beyond fluorinated intermediates, Daikin also provides a series of fluorinating agents.
Daikin is unique in offering iodine pentafluoride, which has rarely been used in fluorination reactions due to the difficulty of controlling reactivity. We have commercialized this technology and are able to offer this low cost, highly selective process to manufacture a variety of organic compounds.
Fluorinating reagents
Direct Fluorination using Elemental Fluorine
This is one of the lowest-cost fluorination processes. Daikin Industries has the technology for controlling the extreme reactivity of elemental fluorine. F2 can be an industrial agent for relatively simple compounds that have a clearly identified reactive site.
Fluorination using IF5/Et3N·3HF Complex
The IF5/Et3N·3HF complex is a unique fluorinating reagent which can fluorinate both electrophilic and nucleophilic sites. Daikin Industries has commercialized this unique technology and is now able to offer this low cost, highly selective process to manufacture a variety of organic compounds.
Fluorination using Sulfur tetrafluoride SF4
Daikin Industries is committed to manufacturing compounds using Sulfur tetrafluoride on a commercial scale. SF4 is a versatile reagent used to convert a carbonyl into a difluoromethyl group or to obtain a trifluoromethyl from a carboxylic acid.
Fluorinated Building Blocks
A wide range of building blocks can be derived from Daikin Industries‘ raw materials (tetrafluoroethylene, hexafluoropropene, hexafluoroacetone, tetrafluorooxetane and perfluoroalkyliodides).
| Chemical name | Chemical formula | CAS NO | TSCA |
| 1H,1H-pentafluoropropanol (5FP) | CF3CF2CH2OH | 422-05-9 | Y |
| 2-(perfluorobutyl)ethanol (C4 Alcohol) | F(CF2)4CH2CH2OH | 2043-47-2 | Y |
| 2-(perfluorohexyl)ethanol (C6 Alcohol) | F(CF2)6CH2CH2OH | 647-42-7 | Y |
| 1H,1H,3H-tetrafluoropropanol | CHF2CF2CH2OH | 76-37-9 | Y |
| 1H,1H,5H-octafluoropentanol | H(CF2)4CH2OH | 355-80-6 | Y |
| 1H,1H,7H-dodecafluoroheptanol | H(CF2)6CH2OH | 335-99-9 | Y |
| 2H-hexafluoro-2-propanol (HFIP) | (CF3)2CHOH | 920-66-1 | Y |
| (perfluorohexyl)ethylene (C6 Olefin) | F(CF2)6CH=CH2 | 25291-17-2 | Y |
| perfluorohexyl iodide (C6 Telomer) | F(CF2)6I | 355-43-1 | Y |
| 2-(perfluorohexyl)ethyl iodide | F(CF2)6CH2CH2I | 2043-57-4 | Y |
| 2-(perfluorohexyl)ethyl methacrylate (C6 SFMA Monomer) | F(CF2)6CH2CH2OCOC(CH3)=CH2 | 2144-53-8 | Y |
| 2-(perfluorohexyl)ethyl acrylate (C6 SFA Monomer) | F(CF2)6CH2CH2OCOCH=CH2 | 17527-29-6 | Y |
| hexafluoroisopropyl methyl ether | (CF3)2CHOCH3 | 13171-18-1 | N |
| 2,2-bis(3,4-anhydrodicarboxyphenyl)hexafluoropropane (6FDA) | C19H6F6O6 | 1107-00-2 | Y |
If you are looking for a fluorinated compound not mentioned in the list, please contact us.
Nucleophilic Fluorinating reagent
Characteristics
- F-81 is a nucleophilic fluorinating reagent with similar reactivity as diethylaminosulfur trifluoride (DAST). F-81 can substitute the hydroxyl group with fluorine under mild conditions.
- F-81 is a source of HF that can be handled much more easily than anhydrous HF, and is a liquid with a high boiling point.
- F-81 and F-82 can both be supplied in bulk.

Structure and properties
| Chemical Name | 1,1,2,3,3,3-Hexafluoro-1-diethylamino-propane |
| Chemical Formula | C7H11F6N |
| Molecular Weight | 223.16 |
| Appearance | colorless transparent liquid |
| Boiling Point | 56°C / 58mmHg |
| Specific Gravity | 1.23 |
| CAS No. | 309-88-6 |
| Chemical Name | Triethylamine trishydrofluorides |
| Chemical Formula | C6H18F3N |
| Molecular Weight | 161.21 |
| Appearance | colorless liquid |
| Boiling Point | 70°C / 15mmHg |
| Specific Gravity | 0.989 |
| CAS No. | 73602-61-6 |
Iodine pentafluoride (IF5) has been rarely used in the fluorination of ordinary organic compounds until recently because of the difficulty in controlling its reactivity. However, Hokkaido University’s Professor Yoneda discovered that the use of IF5 in combination with Et3N·3HF salt significantly improves the reaction selectivity and the process as a low-cost and generally applicable industrial fluorination technology, and is using it in the industrial synthesis of a variety of fluorine-containing organic materials.
An extremely unique aspect of the fluorination using IF5/Et3N·3HF salt is that the fluorinating reaction of organic compounds occurs at both nucleophilic and electrophilic sites as illustrated in the scheme below.

Sulfur tetrafluoride can be used to efficiently fluorinate hydroxyl-, carbonyl and ester groups. DAIKIN Industries is committed to manufacturing compounds using Sulfur tetrafluoride on commercial scale. SF4 is a versatile reagent used to convert a carbonyl into a difluoromethyl group or to obtain a trifluoromethyl from a carboxylic acid, see scheme below.

Electrophilic fluorinating reagent
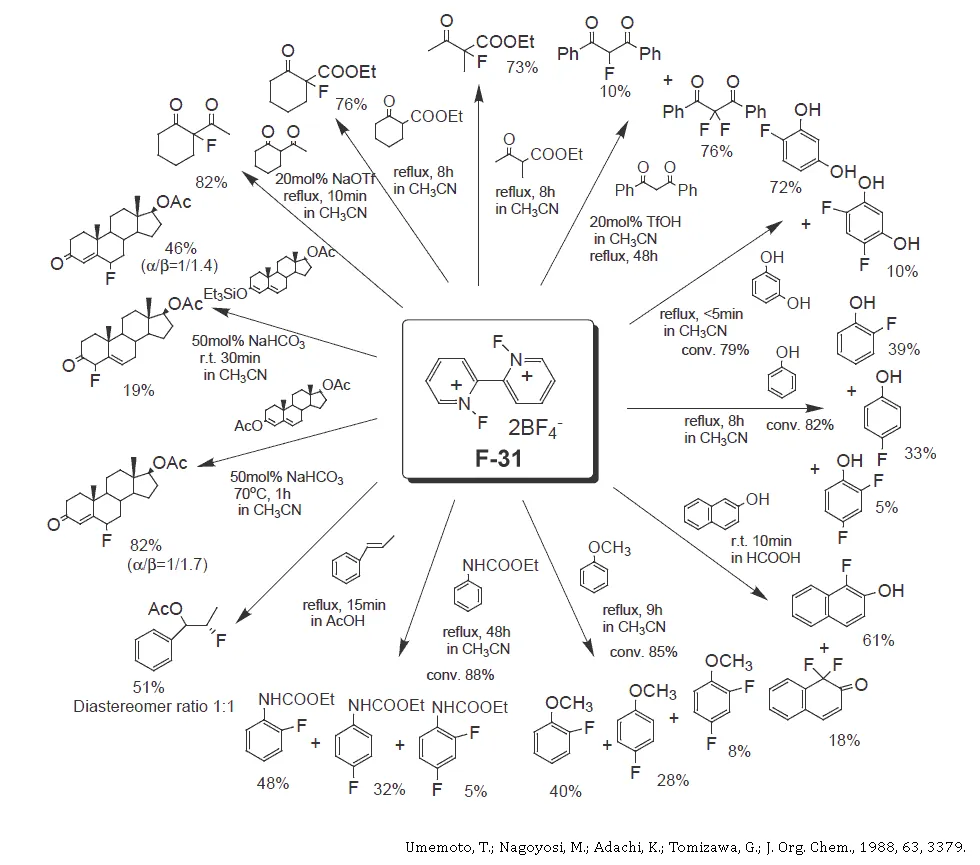
Electrophilic fluorinating reagent with the most effective fluorine content in its class (available in bulk)
Characteristics
- High Effective Fluorine-Content: 103g/kg (Selectfluor: 53.6g/kg)
- Easy to handle; a glass reactor can be used.
- Cost effective fluorinating agent.
- Easy to remove another product, 2,2′-Bipyridinium bis(tetrafluoroborate) form the fluorination products, as it dissolves in diluted hydrochloric acid.
- Wide variety of application in pharmaceutical and agricultural industries, etc.
- Perfect Recycle System.
Structure and properties
| Chemical Name | N,N‘-Difluoro-2,2′-bipyridinium bis(tetrafluoroborate) |
| Mol Formula | C10H8F10N2B2 |
| Mol Weight | 367.79 |
| Appearance | White Crystal |
| Melting Point | 166-168°C (with decomp.) |
| Solubility | Acetonitrile 4.7 mg/ml (25°C) |
| Effective F-Content | 103g/kg (two F+/Mol Weight) |